-

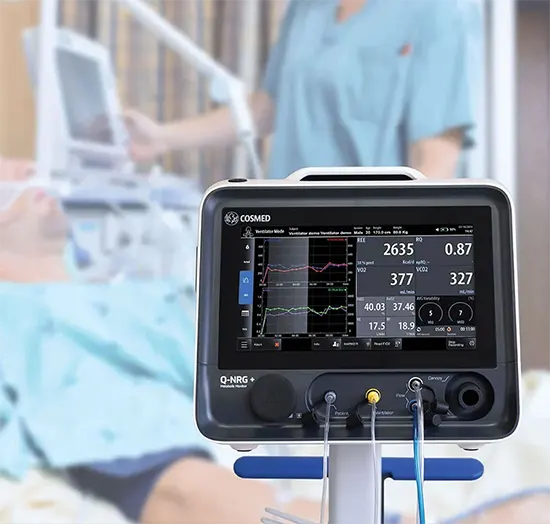
Q-NRG MAX
METABOLIC MONITORIntroducing The New Generation of Metabolic Monitor for VO2 Max and Resting Energy Expenditure
-

-

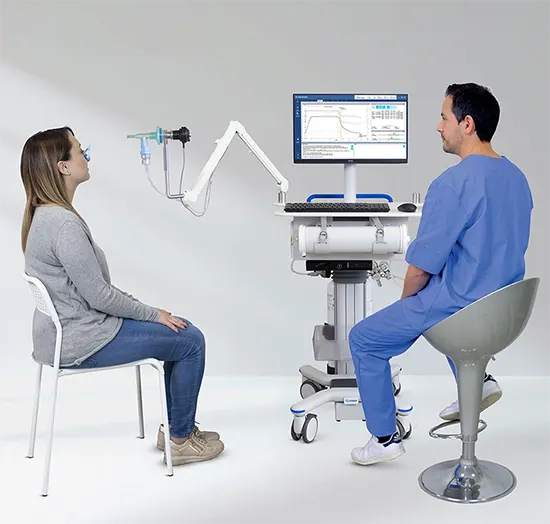
Quark PFT
PULMONARY FUNCTION TESTINGInnovative modularity and networking for truly customised PFT solutions
-
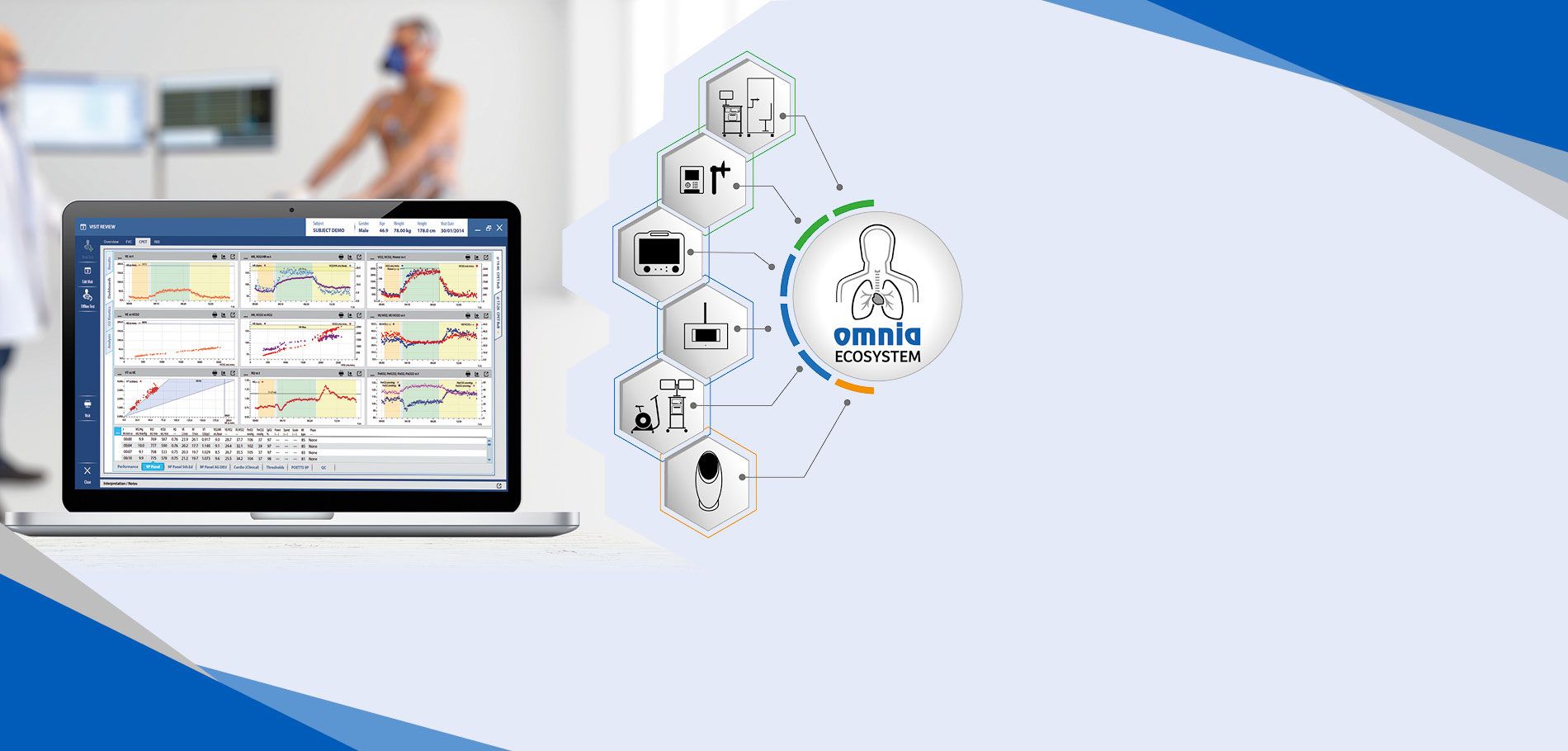
OMNIA Platform
DATA MANAGEMENT SOFTWAREThe most comprehensive Software for Metabolic, Lung Function and Body Composition
COSMED, since 1980, designs innovative solutions for Lung function, Metabolic, and Body Composition Assessment


Years of experience
International Offices
Countries
Products sold
Peer review citations
CARDIO PULMONARY EXERCISE TEST
The most comprehensive CPET Solutions from Clinical to Sport Science
PULMONARY FUNCTION TEST
Modularity and networking for custom Lung Function testing solutions
COSMED Specialities Lines
COSMED exclusive focus on cardiopulmonary, metabolic and body composition measurement technologies,
over the years has brought the company to be considered among the key players of its market



COSMED News
Read the latest news about our Company and discover the upcoming international Events
Get in touch
COSMED strives to provide the best service possible with every contact! Fill the online forms to get the info you're looking for right now!